If 0.0958g of an unknown metal was reacted with HCI, according to the equation below, and 36.5mL of He was collected over water at 23.0☌ and 762mmHg, what is the molar mass of the metal (X)? X (s) + 2 HCl(aq) → XCl2 (aq) + H2 (g) 0. Olgaty PH₂ = 761 mmity V=HRT Р R 0.0821 Theoretical yield: Percent yield: 123Įxperiment 12: Generating Hydrogen Gas 4. (741 mmHg) (1 atm / 760 mmHg) 0.975 atm Use the ideal gas law to find out how many moles of gas were. of hydrogen gas was collected over water at 23☌ and 761 mmHg, calculate the theoretical yield of hydrogen gas (in ml) and finally, calculate the percent yield.
If 0.500g of magnesium was reacted with HCl and 461ml.

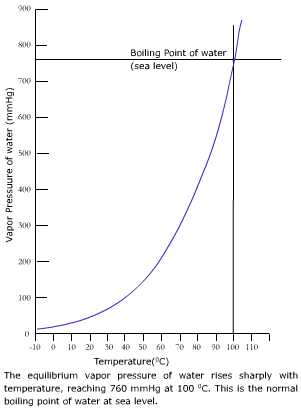
As you know, mole fraction is defined as the number of moles of a component of a solution divided by the total number of moles present in that solution. This means that all you have to do is determine the mole fraction of water in the solution. If the atmospheric pressure is 749mmHg and the gas is collected over water at 25☌, what is the pressure of the hydrogen gas? P = 749 mmHg P H2=P-H₂O = 749-23.8 T= 25C = 23.8mily PH2 = 1725.2mm Hg) na 3. In your case, you know that the vapor pressure of pure water at 25'C' is equal to 23.8 torr.

Transcribed image text: Experiment 12: Generating Hydrogen Gas Prelab: Name: 1.
